Affiliate links on Android Authority may earn us a commission. Learn more.
Study reveals a way to prevent lithium batteries from catching on fire

Lithium batteries are powering most of our devices. From watches, smartphones and tablets to vehicles and even homes. These have proven to be a clean source of energy that is also very efficient, but you do hear of the occasional battery that bursts into flames. This is especially bad when it happens with smartphones, which you carry around and keep dangerously close at all times. Sure, it’s a bit rare for this to happen, but it’s still a factor we need to consider and work on.
Scientists have discovered a way to make lithium batteries much safer in the future. More importantly, it could help the industry speed up the development of lithium-sulfur and lithium-air batteries, which are said to be able to hold about 10 times more energy per weight. Just imagine any phone being able to last about 10 times more energy. We are talking about battery life that could last well over a week. Now, that sounds like a promising future!

What’s the problem with current lithium batteries?
So there seems to be a major set-back in the way lithium batteries currently operate. This discrepancy could also be one of the main reasons why batteries sometimes catch on fire.
Lithium batteries degenerate with time. As the electrode degrades, “fingers” of lithium (otherwise known as dendrites) form in its surface. These can then pierce through the protective separator that keeps the other half of the battery safe. Dendrites getting to the other side can cause issues with short outs, overheating and even fires.
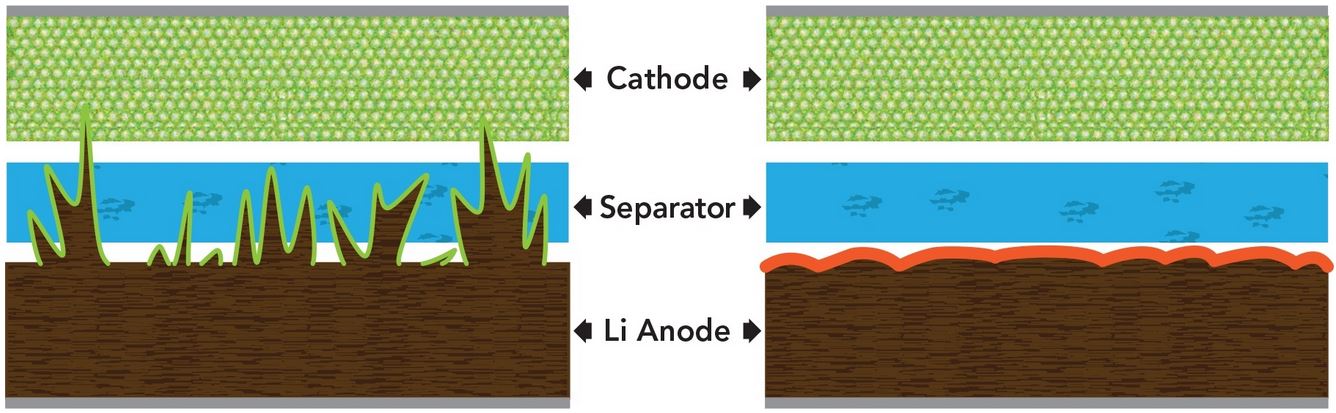
The solution to dendrites in batteries
Doesn’t sound fun, right? There are ways to make “smart batteries” that would detect “fingers” getting out of hand and alert the user, so he may replace it, but that is hardly a solution… it’s more of a security feature.
The method at hand also allows batteries to perform much better in the long run. Tests indicate batteries charged for 150 cycles show significant degradation. By adding these two chemicals to the equation, they were able to charge batteries 300 times and keep 99% efficiency. So not only is this method safer, but it makes batteries much more powerful.
This could bring great changes to our beloved Android smartphones and tablets, but we do need to look further than that. Think about the fact that now smartphones hardly ever come with user-removable batteries. We are not meant to keep these devices for long and usually switch to new ones before the battery degrades significantly. And if you do keep it, there’s no way to easily switch out the battery.
Not all electronics work like that, though. Some products we have to keep longer, like electric cars or home batteries. We wouldn’t want those catching on fire after some time with them, right?
Thank you for being part of our community. Read our Comment Policy before posting.