Affiliate links on Android Authority may earn us a commission. Learn more.
Breakthrough MIT discovery doubles lithium-ion battery capacity

Moore’s law has held strong for processors, making our smartphones exponentially more powerful with every passing year. However, battery technology has proven to be a frustrating barricade. Although our phones are more intelligent about how they manage battery life, we’ve been using essentially the same kinds of batteries for twenty years with only incremental boosts to performance. That is, until now.
Researchers at the Massachusetts Institute of Technology are reporting that they’ve discovered a way to make lithium-ion batteries fully twice as powerful as previous designs. This design is in alignment with current manufacturing procedures, which means these double-charged batteries could be hitting the market as early as next year. Curious as to how it works?
Want to learn more? Here’s a guide on battery-powered motorcycles from Green Authority
In traditional lithium-ion batteries, ions move from a negatively charged graphite cathode to a positively charged anode through an electrolyte solution. Graphite has proven to be an excellent cathode because it’s stable and can hold a relatively high amount of ions.
What would be better than graphite is a lithium-metal foil, which has a far greater capacity for ions. Researchers have known this for some time, but the problem is that lithium-metal has a tendency to react violently with electrolyte solutions, causing overheating and even catching fire.
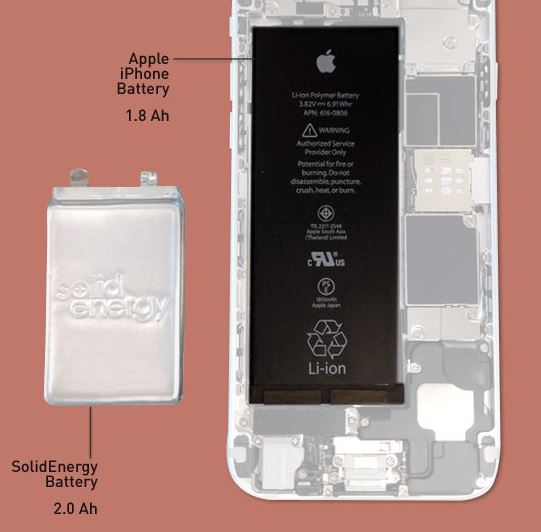
However, researchers with SolidEnergy Systems, a company with its roots in MIT, have developed a new form of electrolyte solution that doesn’t overreact to lithium-metal. With this technology in place, batteries can be made half the size of modern batteries and still retain their full capacity. Alternatively, you can make batteries comparable in size to today’s batteries, and they’ll last you twice as long.
This breakthrough has widespread implications for the mobile market and the electric car industry.
What are the best electric cars? What about the best motorcycles? Find out on Charged.io.
What are your thoughts regarding SolidEnergy’s lithium-metal solution to this decades-long battery problem? Will this be the battery technology of the future? Let us know your take in the comments below!
Thank you for being part of our community. Read our Comment Policy before posting.